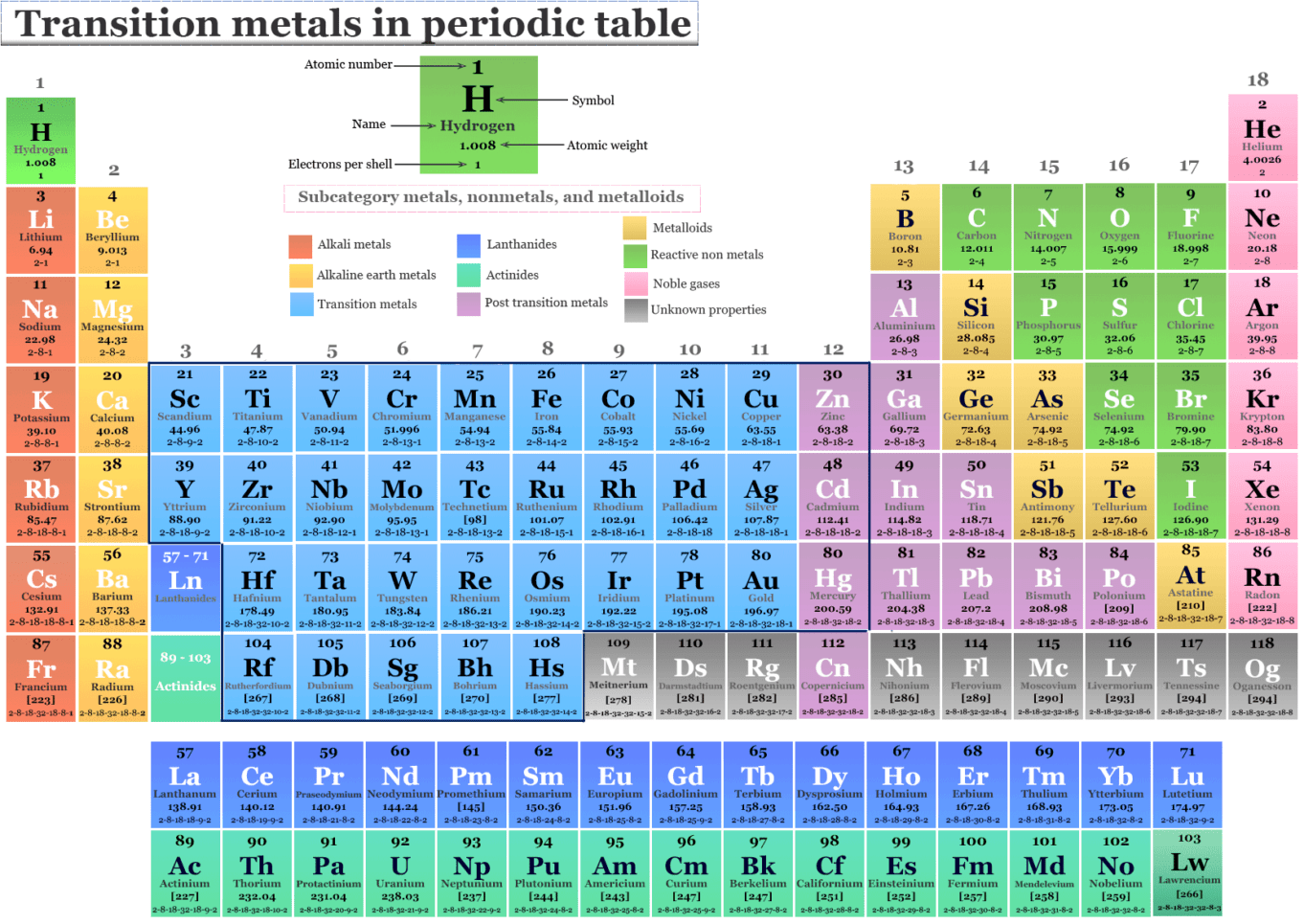
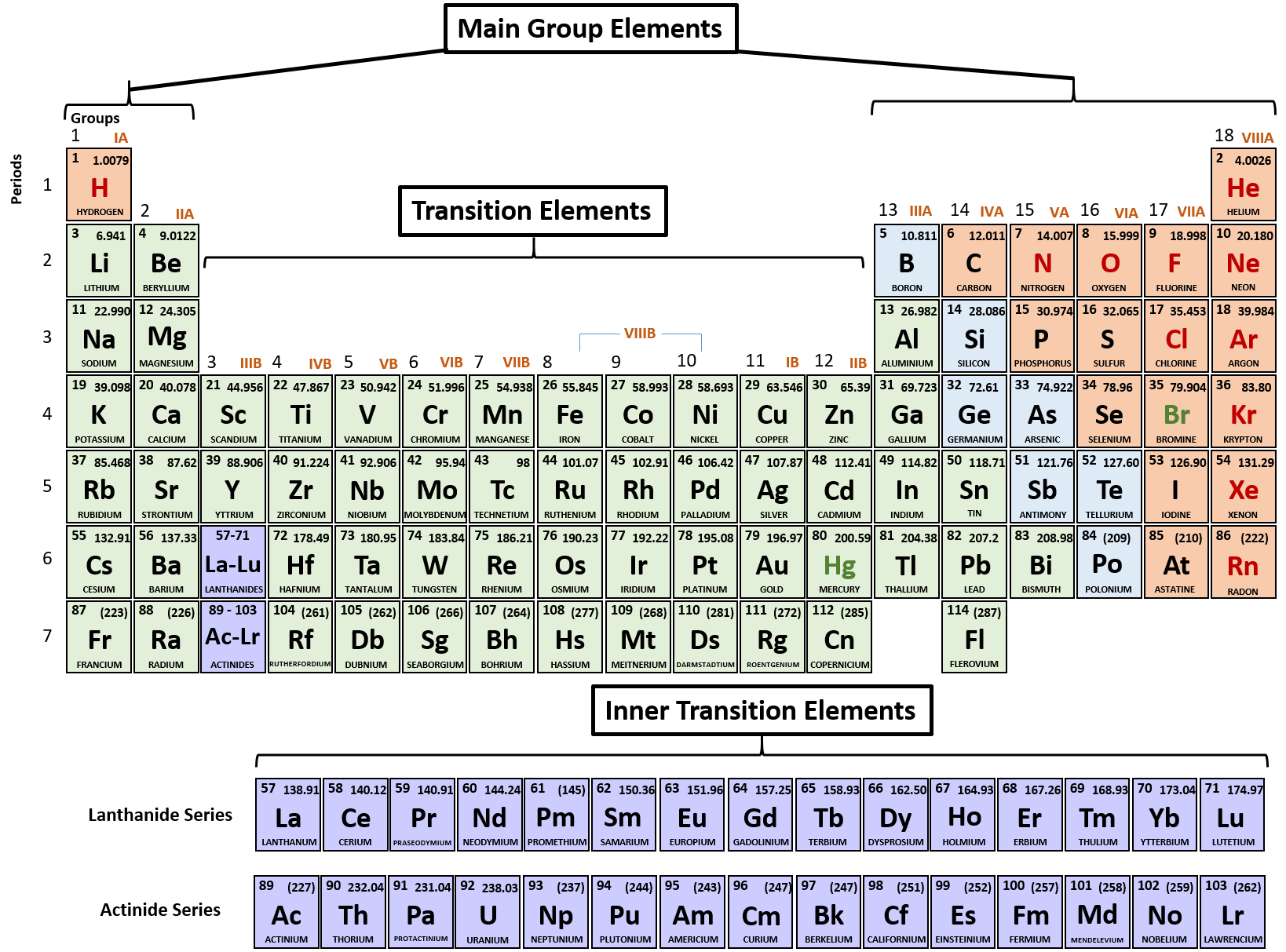
Those two orbitals can use the electrons to bond with other atoms. Example: Molybdenum (Mo), with 42 electrons. Whenever you have a shell that is not happy, the electrons want to bond with other elements. Why can they do that? As you learn more, you will discover that most transition elements actually have two shells that are not happy. It's a chemical trait that allows them to bond with many elements in a variety of shapes. Transition metals can use the two outermost shells/orbitals to bond with other elements. Flashcards in Transition Metals 211 Start learning. They are found between groups 3-12 on the periodic table. Most elements can only use electrons from their outer orbital to bond with other elements. Transition metals are metals whose atoms have a partially filled d-subshell, or which form at least one stable ion with a partially filled d-subshell of electrons. You will find it's usually 2, 8, 18 or 32 for the maximum number of electrons in an orbital. No shell can have more than 32 electrons. Something like gold (Au), with an atomic number of 79, has an organization of 2-8-18-32-18-1. The transition metals are able to put up to 32 electrons in their second-to-last shell. You need to remember that those electrons are added to the second-to-last shells. This is the point in the periodic table where you can place more than 8 electrons in a shell. Scandium (Sc) is only 3 spots away with 21 electrons, but it has a configuration of 2-8-9-2. It has 18 electrons set up in a 2-8-8 order. Transition metals are able to put more than eight electrons in the shell that is one in from the outermost shell. Not all of them, but we are sure you've seen pictures of silver (Ag), gold (Au), and platinum (Pt). At the left of the table is a group that contains reactive. At the right of the table is a group that contains inert gases elements with full valence shells and which are generally unreactive. You will usually find that transition metals are shiny, too. Each row of elements in the periodic table is called a period, and the elements are grouped according to the fill of electrons in their valence shells. They have a lot of electrons and distribute them in different ways. Transition metals are good examples of advanced shell and orbital ideas. We like introducing students to the first eighteen elements, because they are easier to explain. It all has to do with their shells/orbitals.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
